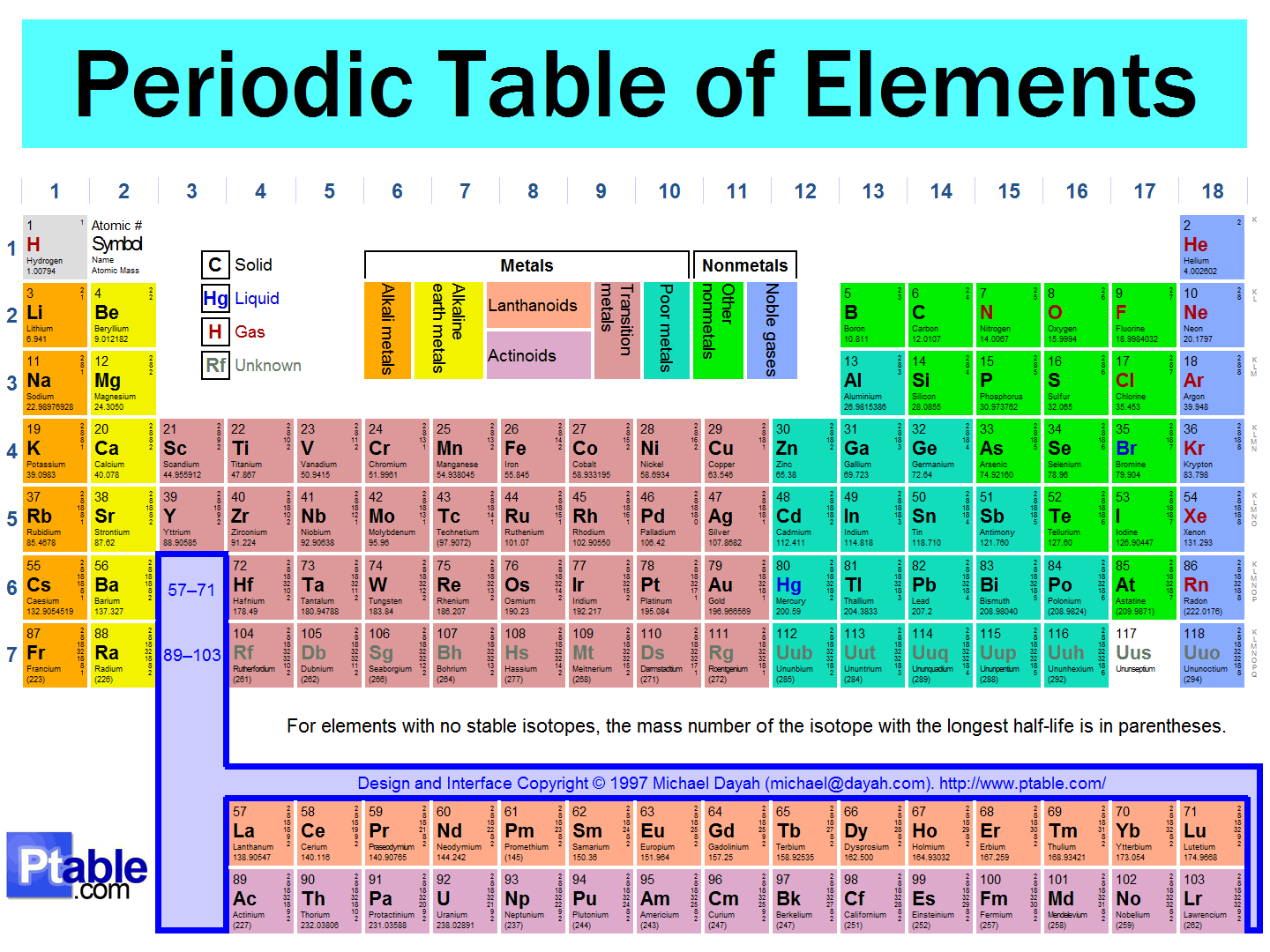
calculate the mass of silver deposited from silver nitrate solution by a current of 2a flowing for 30 minutes - Chemistry - TopperLearning.com | sfx3ujj

Calculate the ratio between the mass of one atom of hydrogen and mass of one atom of silver - CBSE Class 9 Science - Learn CBSE Forum

Silver crystallises in an fcc lattice. The edge length of its unit cell is 4.077 x 10–8 cm and its density is 10.5 g cm-3. Calculate on this basis the atomic mass
![Calculate the mass of: Cong] (i) an atom of silver (Atomic mass of silver = 108 ) ii) a molecule of CO2. with the first orbit in H . Calculate the mass of: Cong] (i) an atom of silver (Atomic mass of silver = 108 ) ii) a molecule of CO2. with the first orbit in H .](https://toppr-doubts-media.s3.amazonaws.com/images/2444574/44e7b4c9-6dd0-49ae-8e9d-b5fb412652aa.jpg)
Calculate the mass of: Cong] (i) an atom of silver (Atomic mass of silver = 108 ) ii) a molecule of CO2. with the first orbit in H .







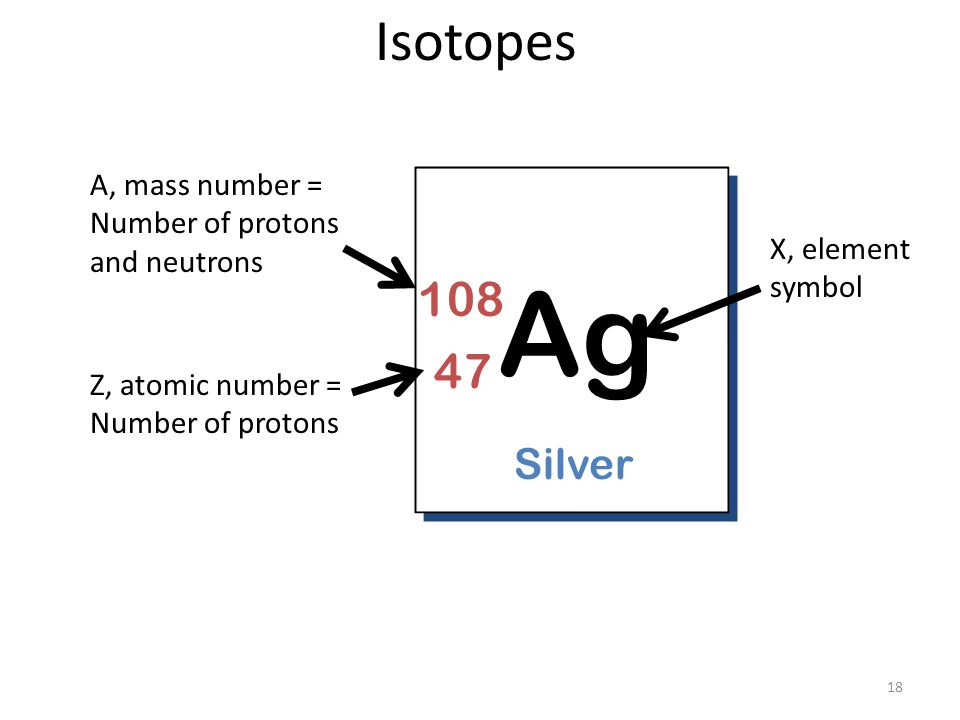
![Silver atom diagram concept - Stock Illustration [50083104] - PIXTA Silver atom diagram concept - Stock Illustration [50083104] - PIXTA](https://en.pimg.jp/050/083/104/1/50083104.jpg)